|
Study expected to include patients with top priority COVID-19 variants, including Delta
Infectious disease company, Appili Therapeutics (TSX: APLI; OTCQX: APLIF), has completed enrollment in its global Phase 3 PRESECO (PREventing SEvere COVID-19) trial evaluating Avigan®/Reeqonus™(favipiravir) as an oral treatment for COVID-19. Appili expects to report top-line data from this trial in approximately 60 days. With today’s announcement, the double-blind, placebo-controlled, randomized, global, multi-center trial has enrolled 1,231 patients from 38 study sites across the United States, Mexico, and Brazil. “Completing enrollment in this large Phase 3 pivotal trial in just ten months is a tremendous accomplishment for Appili. With this major milestone now achieved, we have a clear path to completing the PRESECO data analysis in the coming weeks and determining if Avigan/Reeqonus has the potential to meet the urgent global clinical need for a safe and effective oral treatment for this deadly disease,” said Armand Balboni, M.D., Ph.D., Chief Executive Officer, Appili Therapeutics. The primary goal of PRESECO is to evaluate the impact of oral Avigan/Reeqonus on time to COVID-19 symptom resolution. The study also aims to evaluate the effect of oral treatment on progression to more severe COVID-19 disease. Finally, this Phase 3 clinical trial includes a viral shedding sub-study, which is evaluating impact of the treatment on time to viral clearance. Viral isolates will also be sequenced for identification of COVID-19 variants. “As we have seen over the past 18 months, this virus continues to mutate, enhancing its ability to infect and cause significant symptoms, especially in those who have not been vaccinated. My hope is that Avigan/Reeqonus will emerge as a standard of care treatment for mild to moderate COVID-19 patients, resolve patient’s symptoms more rapidly, and limit progression to more severe disease” said Yoav Golan, M.D., Chief Medical Officer, Appili Therapeutics. About the PRESECO Clinical Trial The Phase 3 PRESECO (PREventing SEvere COVID-19 Disease) study is a double-blind, placebo-controlled, randomized, multi-center, global superiority trial investigating the safety and efficacy of oral Avigan/Reeqonus in the early treatment for adults infected with COVID-19 and showing mild-to-moderate symptoms. Participants were enrolled at multiple clinical trial sites in the United States, Brazil and Mexico. Participants are outpatients with mild-to-moderate symptoms who have had a recent positive COVID-19 test (within 72 hours of enrollment). Participants self-administer the drug regimen in their homes, with clinical investigators monitoring patients remotely. About Avigan/Reeqonus Avigan/Reeqonus is a broad-spectrum antiviral in oral tablet form. It is a selective inhibitor of viral RNA-dependent RNA polymerase (RdRP) with potent antiviral activity against single-stranded RNA viruses, including coronaviruses. Developed by FUJIFILM Toyama Chemical Co., Ltd. (FFTC) and approved in Japan as a treatment and stockpile countermeasure for pandemic influenza. Unlike most other interventions that researchers are evaluating in COVID-19, Avigan/Reeqonus has already been thoroughly studied in human trials and has a well-known safety profile, with over 3,000 subjects receiving at least one dose of the drug. Avigan/Reeqonus’ oral tablet form is shelf-stable and has an established commercial manufacturing process, which may provide advantages over other COVID-19 interventions, which often require temperature-controlled storage and/or injection or intravenous administration. Appili has joined a consortium of companies, including Dr. Reddy’s Laboratories, Global Response Aid, and FFTC, for the worldwide development and distribution of Avigan/Reeqonus tablets for the potential treatment and prevention of COVID-19 (excluding Japan, China, and Russia). This consortium is designed to ensure that Avigan/Reeqonus is thoroughly evaluated in rigorous clinical studies and, if it receives the proper regulatory approvals, has the infrastructure in place to support worldwide manufacturing and distribution. Source: Appili Therapeutics Inc.
0 Comments
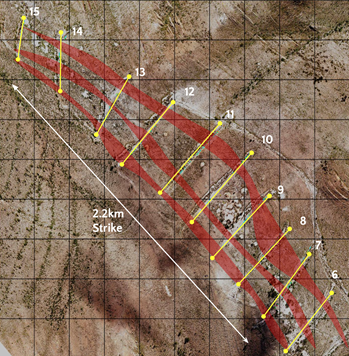
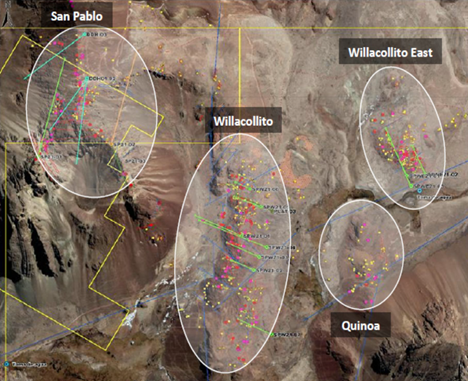
Andean Precious Metals Launches 20,000 Metre Exploration Program and Commences Trading on the OTCQB9/22/2021 Established Latin American silver producer, Andean Precious Metals (TSX-V: APM, OTCQB: ANPMF), has launched a 20,000 metre, phase one exploration campaign focused on the company’s wholly-owned Rio Blanco and San Pablo projects in Bolivia. The mulit-project exploration program is being undertaken to extend the life-of-mine at Andean's flagship San Bartolomé project to ten years. Yesterday, the company also announced that its common shares commenced trading on the OTCQB Venture Market under the symbol “ANPMF”. “Since becoming a public company in March, our focus has been on expanding our existing resource and reserve base along with the scope and profitability of our third party ore purchasing business. We have made significant progress on both fronts and are now well positioned to advance an aggressive exploration campaign. Our portfolio of exploration assets includes two highly prospective gold projects that are within the operating envelope of San Bartolomé. We believe that the San Pablo and Rio Blanco Projects have the potential to create significant value for the shareholders of Andean, and we look forward to updating the market with drill results beginning in October.” - Simon Griffiths, President and CEO of Andean. Phase One Exploration Program Overview Andean Precious Metals controls a portfolio of exploration assets in Bolivia, of which the San Pablo and Rio Blanco Projects (“San Pablo” and “Rio Blanco”) are the most advanced. Both projects are located within the operating envelope of the San Bartolomé Mine, which is currently producing precious metals from six different locations in Bolivia. Rio Blanco and San Pablo are both bulk-tonnage, potentially surface-mineable targets and are being evaluated as synergistic with San Bartolomé. The Company plans to drill 10,000 metres at Rio Blanco and 10,000 metres at San Pablo. Currently, three diamond drill rigs are contracted and active. Once all assays are received from the Phase One exploration program, the Company expects to launch a Phase Two exploration program of an additional 20,000 metres. Both phases are fully funded. Contracts have this week been signed with Quantec for Titan geophysical surveys at both properties. Rio Blanco The 100% owned Rio Blanco Project comprises 13,462 hectares in the department of Potosi. Rio Blanco is located on an historic Au-Sb belt that hosts some of the most prolific mining districts in Bolivia, including Caracota, Chilcobija, Sucre, Candelaria, and Rosa de Oro. Rio Blanco is an Orogenic gold deposit hosted in a folded Ordovician sedimentary sequence composed of shales and sandstones that were affected by a low-grade metamorphism. Extensive geological mapping has been completed and 1500 samples assayed at ALS in Lima. The Phase One diamond drill program covers an initial 7km of a 22km long structural corridor along the axial plane of Rio Blanco anticline. This structural system hosts ore mineralisation in formal veins, stockworks, saddle reefs and lenticular bodies. Rio Blanco has extensive historical workings, including several placer gold deposits. Similar Au-Sb occurrences are well known around the world, such as Bendigo in Australia. Andean’s phase one drill program has 22 diamond drill holes planned which follow an extensive program of geological mapping and trenching perpendicular to the axis of the target mineralisation. San Pablo The 100% owned San Pablo Project comprises 650 hectares in the department of Potosi. San Pablo is located at the southern end of a prolific orogenic gold belt that hosts several of Bolivia’s largest deposits including the Kori Kollo gold mine (Newmont) which produced more than 5 million ounces and the San Bernadino Gold mine which produced over 2 million ounces ounces. The Company has completed initial exploration works including geological mapping, surface sampling and an initial diamond drill programme comprising twelve holes into four discrete targets which are within close proximity of each other. DDH results are expected to be released during October. Early work suggests gold-hosted mineralization occurs near surface in an intrusion-related gold system with narrow sheeted veins together with separate, more discrete veins. The presence of secondary biotite alteration may indicate the presence of porphyry-style mineralization on the property. The Company is planning to test for the presence of porphyry-style mineralization with its new exploration work. Quantec Geoscience's Titan 24 DCIP and magnetotelluric ("DCIP-MT") technology is being considered in a 17 line-kilometre survey. This programme is scheduled for the second half of October. Qualified Person The scientific and technical content disclosed in this press release was reviewed and approved by Donald J. Birak, A Qualified Person as defined by Canadian National Instrument 43-101, Registered Member, Society for Mining, Metallurgy and Exploration (SME), Fellow, Australasian Institute of Mining and Metallurgy (AusIMM). Source: Andean Precious Metals Corp. New Funds Will Help Expedite Completion of Recently Expanded Pivotal Phase 3 Study on Oral Antiviral Medicine
Infectious disease company, Appili Therapeutics Inc. (TSX: APLI; OTCQX: APLIF), has entered into an agreement with FUJIFILM Toyama Chemical Co., Ltd. (“FFTC”), that will provide funding support for its Phase 3 PRESECO (PREventing SEvere COVID-19) clinical trial. The new funding of $1,000,000 USD was secured from FFTC, one of the Company’s partners in a global consortium focused on the worldwide development, commercialization, and distribution of Avigan®/Reeqonus™ (favipiravir) tablets for the potential treatment and prevention of COVID-19. The new funding will support various components of the recently expanded PRESECO trial, including study close-out activities as the Company prepares for database lock and top-line results. Enrollment targets for PRESECO and the viral shedding sub-study were recently increased to enable additional enrollment of COVID-19 variant cases and maximize the number of patients in the final analysis. As part of the agreement, FFTC will receive direct access to PRESECO data in support of local regulatory submissions in Japan. PRESECO is investigating the safety and efficacy of Avigan/Reeqonus in the early treatment outpatient setting for adults infected with COVID-19. “FFTC recognizes the unmet need of this pandemic, which is the lack of safe, effective oral antiviral medicines to treat mild-to-moderate COVID-19 patients. With FFTC’s support, we are one step closer to determining if Avigan/Reeqonus will meet this need,” said Dr. Armand Balboni, Chief Executive Officer, Appili Therapeutics. Avigan/Reeqonus is a broad-spectrum antiviral in oral tablet form. It is a selective inhibitor of viral RNA-dependent RNA polymerase (RdRP) with potent antiviral activity against single-stranded RNA viruses, including coronaviruses. Developed by FFTC and originally approved in Japan as a treatment for pandemic influenza, this product is one of the few oral antiviral candidates in Phase 3 clinical development to treat newly diagnosed COVID-19 patients. “We share the sense of urgency that so many physicians and nurses have expressed globally for the need of new treatments developed that will help bring this pandemic to an end. We remain confident Avigan/Reeqonus will play an important role in North America, Japan, and around the world in the treatment of COVID-19 now, and in the near future,” said Junji Okada, President, FUJIFILM Toyama Co., Ltd. About the PRESECO Clinical Trial The Phase 3 PRESECO (PREventing SEvere COVID-19 Disease) study is a double-blind, placebo-controlled, randomized, multi-center superiority trial investigating the safety and efficacy of Avigan/Reeqonus in the early treatment for adults infected with COVID-19 and showing mild-to-moderate symptoms. Investigators are enrolling participants at multiple clinical trial sites in the United States, Brazil and Mexico. Participants are outpatients with mild-to-moderate symptoms who have had a recent positive COVID-19 test (within 72 hours of enrollment). Participants self-administer the drug regimen in their homes, with clinical investigators monitoring patients remotely. About Avigan/Reeqonus Avigan/Reeqonus is a broad-spectrum antiviral in oral tablet form. It is a selective inhibitor of viral RNA-dependent RNA polymerase (RdRP) with potent antiviral activity against single-stranded RNA viruses, including coronaviruses. Developed by FUJIFILM Toyama Chemical Co., Ltd. (FFTC) and approved in Japan as a treatment and stockpile countermeasure for pandemic influenza. Unlike most other interventions that researchers are evaluating in COVID-19, Avigan/Reeqonus has already been thoroughly studied in human trials and has a well-known safety profile, with over 3,000 subjects receiving at least one dose of the drug. Avigan/Reeqonus’ oral tablet form is shelf-stable and has an established commercial manufacturing process, which may provide advantages over other COVID-19 interventions, which often require temperature-controlled storage and/or injection or intravenous administration. Source: Appili Therapeutics Inc. Ceres Global Ag Corp. (TSX: CRP) today announced its financial and operating results for the quarter and year ended June 30, 2021. All amounts are in U.S. currency unless otherwise noted. Highlights for the quarter and year ended June 30, 2021
Highlights Subsequent to Quarter End
"A strong fourth quarter capped a year of meaningful progress against our broader strategy to partner with growers and other suppliers to enable our downstream customers to do great things in the food, agriculture and energy markets. In fiscal year 2021, we handled 14.4 million more bushels, we strengthened our presence in crucial geographies on both sides of the U.S. and Canadian border, and we maintained the positive trajectory of improved financial performance that began several years ago. Most notably, the profitable execution of our strategy and positive outlook enabled us to recognize $9.7 million in previously unrecognized deferred tax assets during the quarter." - Robert Day, President and CEO of Ceres Global Ag. Outlook Mr. Day continued, "Fiscal year 2022 is shaping up to be a transformative year for Ceres, resulting from years of hard work and exciting growth projects targeted at advancing our strategy. Due to disciplined analysis of markets and prudent acquisition of supplies for key customers, we expect the first quarter of fiscal year 2022 will be a record for Ceres in terms of EBITDA and pre-tax profits. Meanwhile, we are making progress on the canola crush project at Northgate as we prepare to break ground in 2022; Farmers Grain, LLC in Thief River Falls is off to a strong start and the expansion is on track for completion prior to harvest 2022; and the expansion of our soybean crush plant in Jordan, Manitoba, which is positioned to benefit from the fast-growing demand from food, feed, and renewable fuel markets, was completed in July 2021 and is expected to generate increased margins and profits once the new crop harvest begins in October." Conference Call Details Management of Ceres will host a conference call today, September 10, 2021, at 9:00 am ET to discuss its third quarter financial and operational results. All interested parties can join the conference call by dialing 1-888-664-6392 or 416-764-8659. Please dial in 15 minutes prior to the call to secure a line. A live audio webcast of the conference call will be available at: https://bit.ly/CeresQ42021. Please connect at least 15 minutes prior to the conference call to ensure adequate time for any software download that may be required to join the webcast. An archived replay of the webcast will be available for 90 days. Non-IFRS Financial Measures EBITDA (Earnings before Interest, Taxes, Depreciation and Amortization) is not a standardized financial measure prescribed by IFRS; however, it is a metric that is used by management to determine the Corporation's ability to service its debt and finance capital. In calculating EBITDA, Ceres excludes gains and losses on property, plant and equipment, assets held for sale, and gains and losses on equity investments as these items are considered to be non-reoccurring in nature. Ceres may calculate EBITDA differently than other companies; therefore, Ceres' EBITDA may not be comparable to similar measures presented by other issuers. Investors are cautioned that EBITDA should not be construed as an alternative to net income or loss, or to other standardized financial measures determined in accordance with IFRS and is not intended to represent cash flows or results of operations in accordance with IFRS. About Ceres Global Ag Corp. (ceresglobalagcorp.com) Ceres and its subsidiaries ("Ceres Global") add value across agricultural, energy and industrial supply chains through efficient sourcing, storing, transporting and marketing of high-quality agricultural commodities, value-added products and raw materials. Leveraging its network of commodity logistics centers and team of industry experts, Ceres Global connects farmers to customers around the world. Ceres Global is headquartered in Minneapolis, Minnesota, and together with its affiliated companies, operates 13 locations across Saskatchewan, Manitoba, Ontario, and Minnesota. These facilities have an aggregate grain and oilseed storage capacity of approximately 31 million bushels. Ceres Global has a 50% interest in Savage Riverport, LLC (a joint venture with Consolidated Grain and Barge Co.), a 50% interest in Farmers Grain, LLC (a joint venture with Farmer's Cooperative Grain and Seed Association), a 50% in Gateway Energy Terminal (an unincorporated joint venture with Steel Reef Infrastructure Corp.), a 25% interest in Stewart Southern Railway Inc. (a short-line railway located in southeast Saskatchewan with a range of 130 kilometers), and a 17% interest in Canterra Seed Holdings Ltd. (a Canada-based seed development company). For more information about Ceres Global, please visit www.ceresglobalagcorp.com. |
Follow @SharePitch on TwitterArchives
January 2024
Categories |




 RSS Feed
RSS Feed
